NASA Knows: The Ozone Hole
This is the story of the hole in Earth's protective ozone layer: what it is, how it formed, and the decades-long effort to repair it.
Why is there an ozone hole?
Ozone 101 shows the ABCs of CFCs and the atmospheric problem they created.
Watch on YouTubeKey takeaways
- The ozone layer shields Earth from ultraviolet (UV) radiation from the Sun that can harm plants, animals, and humans. The ozone layer forms in the stratosphere 7-31 miles (11–50 km) above the Earth's surface.
- Chlorofluorocarbons (CFCs) and halons are human-produced chemicals, used in products in refrigerants, spray cans, and fire extinguishers. The breakdown of these chemicals causes a reaction with ozone molecules that thins the ozone layer over the poles, most notably Antarctica.
- An international decision to phase out CFCs has slowly reversed the trend of thinning ozone. The Montreal Protocol, an international partnership begun in 1987 with signatories from around the world, controlled the consumption and production of ozone-depleting substances, so that the levels of these have stopped growing in the atmosphere.
- If the progress caused by the Montreal Protocol and subsequent amendments continues, the Antarctic ozone hole could be restored to 1980 levels by 2070.
- NASA and its partners—government agencies, universities, and private sector entitites—develop and operate systems to measure ozone in the atmosphere, using numerous optical methods, which measure the intensity of UV light as it passes through the atmosphere.
The basics
-
What is ozone?
Oxygen exists in two forms in the atmosphere: O2, a molecule with two oxygen atoms, is the air we breathe, and it makes up 21% of the Earth's atmosphere; and O3, or ozone, a molecule with three oxygen atoms, makes up 0.001% of the atmosphere. Ozone is created when solar UV radiation breaks down oxygen molecules in our air.
UV radiation is more intense at higher altitudes because it has not yet passed through much of the atmosphere, so it hasn’t yet been absorbed. The ozone layer forms in Earth’s stratosphere, about 7 to 31 miles (11 to 50 km) above the surface. Ozone also forms at the surface, where it is a harmful pollutant. This drawing shows the chemistry of the ozone generation process.NASA/ Karen Romano Young
This drawing shows the chemistry of the ozone generation process.NASA/ Karen Romano Young -
What does the ozone layer do for Earth?
The UV index is a measurement of the strength of the UV rays reaching Earth’s surface from the Sun. UV is measured on a scale of 1 to 11+, in which 1 indicates a low risk of overexposure and 11+ means a high risk to humans and the environment. Too much UV light can lead to sunburns, skin cancer, and cataracts in humans. It can reduce yields from our farm crops and harm aquatic plants and animals. Ozone controls the UV level, acting as a sunscreen for our planet. It absorbs UV-B—the most harmful form of UV radiation that reaches Earth’s surface—which can damage DNA in plants and animals.
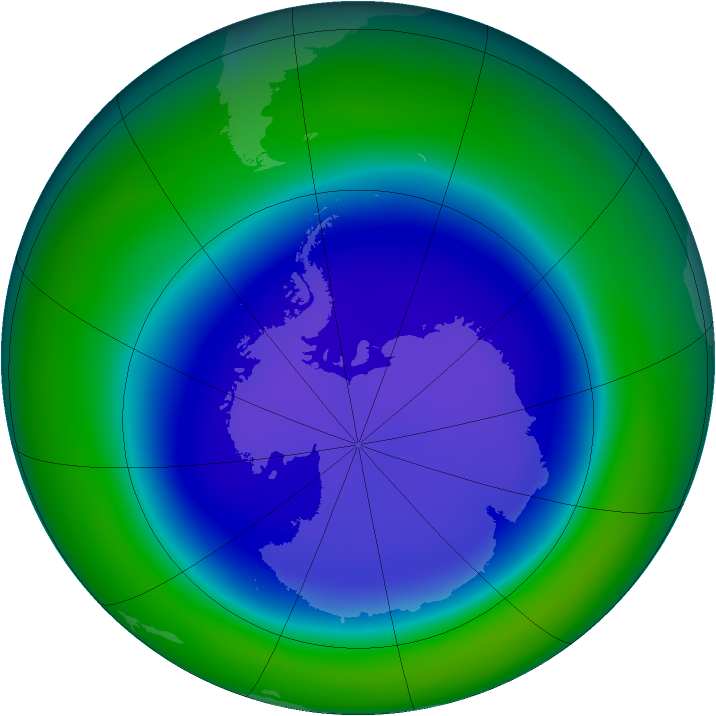
The ozone layer in Earth’s atmosphere is critical to healthy life on Earth. Thin places exist at both poles, but the "hole" is bigger in the Antarctic (see the Arctic, at left). Losing ozone there is such an enormous threat that, when scientists discovered that the ozone layer was thinning, they prompted the international community to act collectively in response to the news, enacting the Montreal Protocol to curb the chemicals that were causing the problem. There is an occasional thinning of the ozone layer in the Arctic, but it poses less risk than the Antarctic ozone hole.NASA’s Goddard Space Flight Center
There is an occasional thinning of the ozone layer in the Arctic, but it poses less risk than the Antarctic ozone hole.NASA’s Goddard Space Flight Center -
What is the ozone hole?
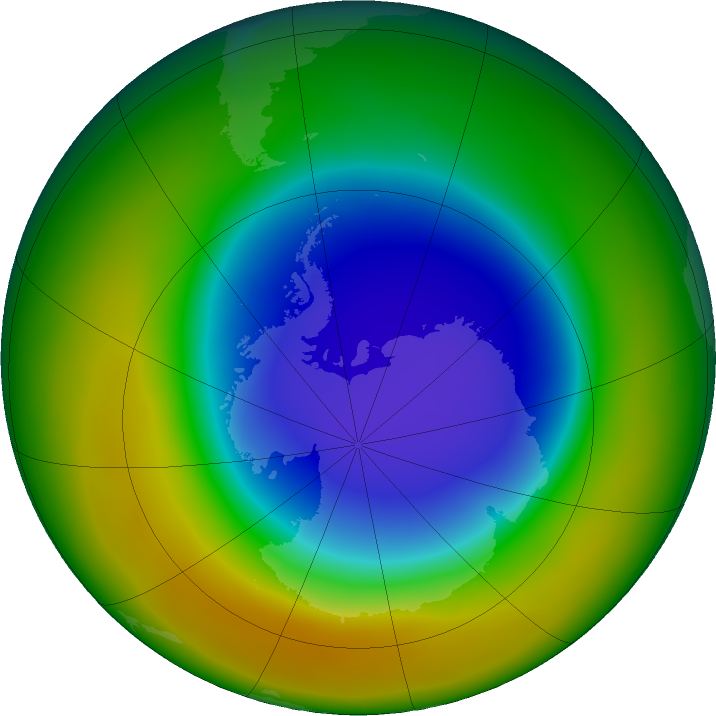
The ozone “hole” is not a hole, but rather a thin patch of depleted ozone in the stratosphere over Antarctica. This thinning is caused by molecules that result from the breakdown in CFCs and other ozone-depleting compounds. These include chlorine and bromine, which are so inert (nonreactive) that they can stay in the atmosphere long enough to be carried up to the stratosphere, where they react with polar cloud particles that ultimately deplete ozone.
 This drawing shows how CFCs divide and react to break down ozone, creating the hole.NASA/Karen Romano Young
This drawing shows how CFCs divide and react to break down ozone, creating the hole.NASA/Karen Romano Young
Vital info
Where is the ozone layer?
The ozone layer forms in the stratosphere 7-31 miles (11-50 km) above Earth, and encompasses the entire planet. Most stratospheric ozone is produced at tropical latitudes, and high-altitude winds spread it over the planet. It is constantly forming and breaking down. Distribution is not uniform. In different places around the world, there are seasonal and long-term variations in the quantity of stratospheric ozone. But over the long run, the ozone layer was stable until humans began producing chemicals that depleted it.
Timing is everything
One of those seasonal variations occurs over the poles. In the dark, sunless winter at high latitudes, a jet stream encircles and isolates cold air. The cold air within this polar vortex enables stratospheric clouds to develop, and these clouds create conditions where chlorine can break free from CFC molecules. Stratospheric clouds provide the surfaces needed for these reactions to take place and, once the sun rises, allow for chlorine and bromine to react with ozone.
An ongoing process
The chlorine atoms that are freed when UV radiation meets CFCs cause reactions that destroy ozone and return the chlorine atoms to the atmosphere unchanged, where they can shred more ozone molecules–as long as the air in the polar vortex remains cold. As the stratosphere warms up, the vortex breaks down. Within a few days of warming, ozone depletion ends, and the ozone layer forms again.
Can stability return?
The ozone concentration significantly lowers throughout winter and early spring until warming air from surrounding latitudes mixes into the cold center of the polar vortex and weakens it. As sunlight returns, UV light starts making new ozone while shutting down the processes that led to its destruction. The ozone-destroying forms of chlorine disperse, and the ozone layer stabilizes until next year.
Is there just one ozone hole?
During winter in Antarctica, the ozone concentration drops because the chlorine from broken-down CFCs destroys ozone faster than it can be made, creating the ozone hole we have today. The same process happens in the Arctic, but because the stratosphere there is warmer, high-altitude clouds don’t form as often or spread as far.


Ozone Hole Maximum to Modern
Ozone hole progress
September 2006 - September 2025
How the science happens
-
How do we measure the ozone hole?
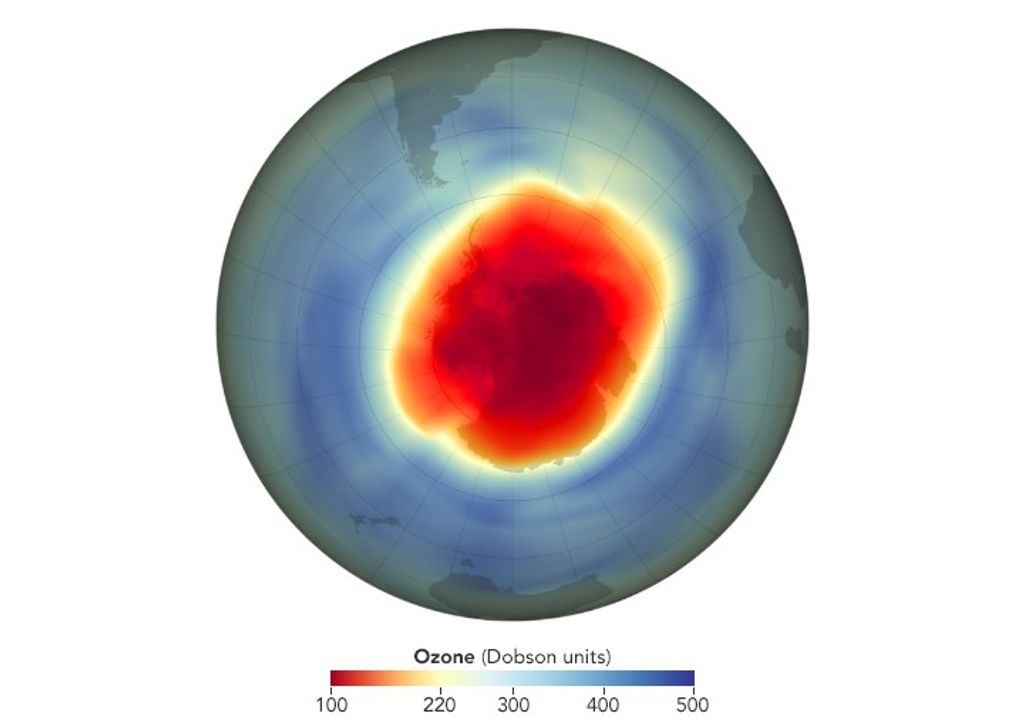
Ozone is often measured in Dobson units (DU). A satellite or aircraft spectrophotometer is aimed straight down to capture the amount of ozone in a vertical column. Think of it like a cylindrical beam of light from space to Earth. For the sake of the measurement, imagine that the ozone were dust, and it all settled to the bottom of the light shaft. This is what Dobson units measure: how much ozone is in the whole column, as if it had settled onto the Earth below. Scientists consider anything below 220 DU thin enough to qualify as a hole.
The area of the ozone hole is found by calculating the area of the Earth covered by less than 220 DU of ozone. Since the ozone hole was discovered in the 1980s, it has been monitored continuously using both ground-based and satellite-based techniques. The ozone layer reaches its lowest point — the ozone hole’s maximum — each year in September and early October, which is the end of winter over the South Pole.
The area of the ozone hole continued to grow for many years. According to satellite records, the thinnest ozone layer — the deepest “hole”— occurred in 1994, when parts of the Antarctic stratosphere had just 73 DU of ozone. The largest hole was observed in 2006. Compressed to sea-level pressure, the ozone in the atmosphere would form a layer about the height of two pennies stacked together. Levels in the ozone hole are much less—only the height of a single dime.NASA/Karen Romano Young
Compressed to sea-level pressure, the ozone in the atmosphere would form a layer about the height of two pennies stacked together. Levels in the ozone hole are much less—only the height of a single dime.NASA/Karen Romano Young
How does NASA help?
- NASA and its partners develop and operate satellite systems that include spectrophotometers, which measure the intensity of UV light as it passes through the atmosphere, revealing the thickness of the ozone layer.
- International collaborators in the Network for the Detection of Atmospheric Composition Change (NDACC) and U.S. National Oceanic and Atmospheric Administration (NOAA) gather ozone data from the ground in Antarctica.
- Additional spectrophotometric data are acquired from space by the Ozone Monitoring Instrument (OMI) on NASA’s Aura satellite and the Ozone Mapping and Profiler Suite (OMPS) on the NOAA-20, NOAA-21 and the NOAA-NASA Suomi NPP. The total column ozone, captured by OMI, is a vertical measure upward from Earth to space. Similar downward-looking measurements come from OMPS, which also takes side-on profiles of the ozone layer.
- NASA’s Global Modeling and Assimilation Office (GMAO) uses high-resolution satellite observations to pull together representations of global stratospheric ozone conditions. These calculations allow scientists to predict what stratosphere-level ozone will do in the decades ahead.
How NASA sees the ozone hole
Watch to learn about NASA's ongoing contributions to the world's understanding of the ozone hole.
Watch on YouTubeResources
Related Missions
Aura – Launched in 2004, Aura carries the Ozone Monitoring Instrument (OMI).
SUOMI NPP – Launched in 2011, the Suomi National Polar-orbiting Partnership is a joint mission between NASA and NOAA to monitor ozone and other environmental variables. The Ozone Mapping and Profiler Suite (OMPS) aboard senses the ozone layer and tracks its recovery. The two successors to Suomi NPP also carry the OMPS instrument.
NOAA-20 – Launched in late 2017, NOAA-20 continues long-term global ozone data records.
NOAA-21 – Launched in late 2022.
TEMPO – Measures air quality over the Northern Hemisphere
GMAO Ozone – NASA’s Global Modeling and Assimilation Office (GMAO) operates the Goddard Earth Observing System (GEOS) Composition Forecasting (GEOS-CF) system
Related Instruments
Spectrophotometer – This instrument measures the intensity of a light beam at different wavelengths, to see how much light is absorbed by the subject. In order to measure ozone in the atmosphere, a spectrophotometer measures the intensity of ultraviolet light absorbed by ozone molecules.
Weather balloon – Weather balloons—which measure atmospheric factors such as temperature, humidity, and pressure—can also carry ozonesondes, which gather data that reveal ozone distribution and concentration in the atmosphere.
Definitions
Chlorofluorocarbons (CFCs)
CFCs were produced in refrigeration, air conditioning, the spray cans for such substances as spray paint, hair spray, and anti-perspirant, and in foam packaging and insulation.
Dobson units (DU)
Dobson units: the amount of ozone present in the column of overlying atmosphere.
Ozone (O3)
A molecule composed of three oxygen atoms.
Stratosphere
The layer of the atmosphere 10–50 km (8 to 30 miles) above Earth’s surface.